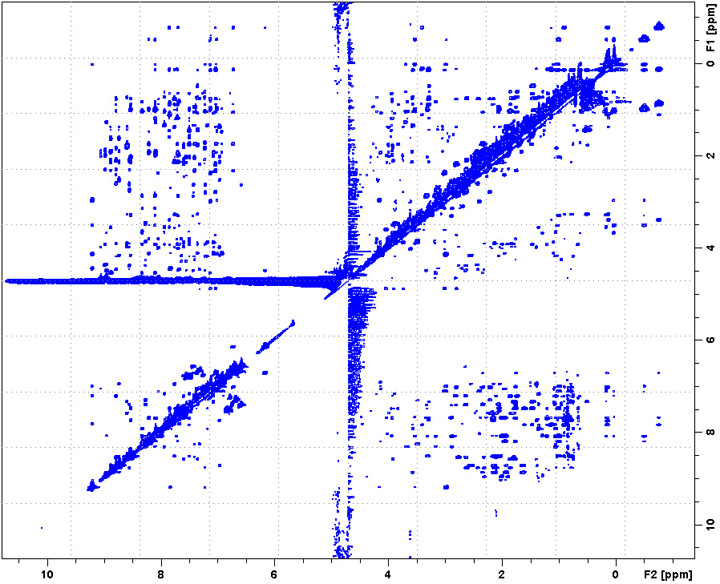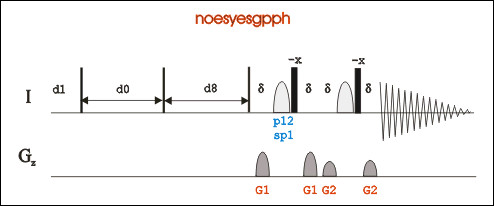
Go to an existing 2D-Noesy experiment (pulse program: noesyesfbgpph), type “edc” to copy its parameters and generate a new experiment number. Or in a new experiment, "rpar noesyesfbgpph".
*Pulse program noesyesfbgpph is based on noesyesgpph and have f2 and f3 channels included.
-
Type "edasp" to check and adjust spectrometer routing.
-
Type “eda”, set parameters td, ns, ds, sw, O1P, O2P and O3P if necessary.
-
In acquisition window, if sample is unlabeled, leave ZGOPTNS blank. If sample is N15 labeled, set ZGOPTNS "-DLABEL_N". If sample is N15 and C13 labeled, set ZGOPTNS "-DLABEL_CN".
-
Type “ased”, adjust power length and power level for 1H. If sample is labeled, you will need to check 15N and/or 13C power level and power length according. Or type "getprosol 1H A B" to set power related parameters. A is the pulse length in µs, and B is the power leverl in dB.
-
Set d8 (NOESY mixing time) around 90 ms for biomolecules, or 300-500 ms for small molecules; set pL32 70dB for water presaturation.
Users may consider using 2D-ROSEY for medium size molecules, MW 1000-3000 Da, mixing time 200ms. -
In gs mode, adjust O1P and p12 to achieve minimum FID area integral for water suppression.
-
Start the experiment by rga and zg.
-
Process the FID by typing “xfb”. Phase the spectrum when necessary.