
Education
- M.S. in Physics, University of Kazan, Russia, graduated summa cum laude 1978
- Ph.D. in Physics and Math.Sci., University of Kazan, Russia 1985
- 1985-1991Scientist, Mol. Biophys. Lab, Kazan Inst. of Biology, USSR Acad. Sci., Russia
- 1991-1993 Alexander von Humboldt Fellow, Institute of Biophysical Chemistry, University of Frankfurt, Germany
- 1993 Research Associate, Inst. of Biophysical Chemistry, University of Frankfurt,Germany
- 1994–2000 Research Associate, Physical Biochemistry Lab, The Rockefeller University, New York
Professional Experience
- Professor, Department of Chemistry and Biochemistry, University of Maryland, 2008-present
- Associate Professor, Department of Chemistry and Biochemistry, University of Maryland, 2005-2008
- Research Associate Professor, Dept Chemistry and Biochemistry, University of Maryland, 2000-2005
- Research Associate, The Rockefeller University, New York, NY, 1994-2000
- Research Associate, Inst. Biophysical Chemistry, University of Frankfurt, Germany, 1993
- Alexander von Humboldt Fellow, Inst. Biophys. Chem., University of Frankfurt, Germany, 1991-1993
- Scientist, Molecular Biophysics Lab., Kazan Inst. of Biology USSR Acad. Sci., USSR, 1985-1991
- PhD Student, Dept. of Theoretical Physics, Kazan State University, Kazan, USSR, 1981-1984
- Teaching/Research Assistant, Dept. Theoretical Physics, Kazan State University, USSR, 1978-1981
- Visiting Professor, CERM, University of Florence, Italy, 2002
- Visiting Professor, Technion — Israel Institute of Technology, Haifa, 2008-2015
- Guest Researcher, NIST Center for Neutron Research, 2015-present
Research Interests
Structure, dynamics, stability, and function of biological macromolecules. Structural biology and molecular mechanisms of intracellular signaling. Ubiquitin-mediated signaling pathways. Structure and dynamics of multidomain proteins. Development and application of new robust theoretical and experimental approaches to protein structure and dynamics, including conformational ensembles, in order to understand their roles in protein stability and function.
Major Recognitions and Honors
- 1986 Award for the best scientific paper of the year 1985 in Physics, by the Ministry of the Highest and Special Secondary Education of the USSR
- Alexander von Humboldt Fellowship Award (1991-1993)
- 2000-2002 Special Grant in the Chemical Sciences from the Camille and Henry Dreyfus Foundation
- Elected Program co-Chairman of the 10th Chianti Workshop on Magnetic Resonance “Nuclear and Electron Relaxation” (Italy, 2003).
Significant Professional Service and Activities
Co-Organizer of the joint NIH & UMD practical training course “Structure determination of biological macromolecules by solution NMR”, 2008, co-Chairman of the 10th Chianti Workshop on Magnetic Resonance “Nuclear and Electron Relaxation” (2003, Italy) and the 12th Chianti/INSTRUCT Workshop on bioNMR (2012, Italy)
Students Mentored
To date thirteen undergraduate and twenty graduate students are co-authors of research publications with Fushman (since 2000). Fourteen postdoctoral associates; sixteen students received Ph.D. degrees, and six students received M.S. degrees.
Structure, dynamics, and function of biological macromolecules; Intracellular signaling; Ubiquitin signaling system
My primary scientific interest is in understanding the relationship between structure, dynamics, and function in complex molecular systems, like proteins. Our research spans molecular biophysics, nuclear magnetic resonance (NMR), small-angle neutron and X-ray scattering (SANS/SAXS) biochemistry, and structural biology: The major focus is on two areas:
(1) structure and dynamics characterization of multidomain systems with the focus on the molecular mechanisms of the ubiquitin signaling system;
(2) development and application of new, robust theoretical and experimental approaches to protein structure and dynamics in order to understand their roles in protein stability and function.
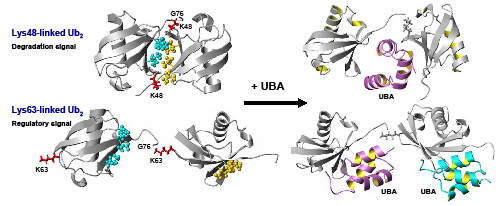
Our research on intracellular signaling is focused on the ubiquitin-proteasome proteolytic system, the principal regulatory mechanism for the turnover of short-lived proteins in eukaryotes. We uncover the structural determinants of the molecular mechanisms involved in the recognition and processing of the ubiquitin signal and allow differently-linked poly-ubiquitin chains to act as distinct cellular signals (see Polyubiquitin chains: polymeric protein signals, by C. Pickart & D. Fushman, Current Opinion in Chemical Biology (2004) 8, 610-616). We have demonstrated that the conformation of poly-ubiquitin chain depends on the linkage, and this provides the mechanism for differential recognition of alternatively linked chains. Our studies focus on the role of the structure and dynamics of poly-ubiquitin signal in its recognition by various receptors.
We are developing new, robust theoretical and experimental approaches to protein structure and dynamics. Our success in answering the challenging questions in protein signaling rests on the improvements in the existing and development of new methodology. We have developed accurate and efficient methods for determining the overall rotational diffusion tensor of a protein from spin-relaxation measurements and for predicting this tensor directly from protein’s structure. We have designed new experimental methods to measure 15N relaxation and the effects of relaxation interference due to cross-correlation between 15N chemical shift anisotropy (CSA) and 1H-15N dipolar interactions. We are developing a comprehensive approach, based on multiple-field measurements, that allows accurate analysis of protein dynamics and accounts for both rotational anisotropy and CSA variability. We have developed methods for structure characterization of multidomain proteins that rely on long-range structural information (rotational diffusion and/or molecular alignment) and provide information on the relative positioning and orientation of the individual domains within a molecule or complex. We have developed new approaches to determine and characterize conformational ensembles of multidomain systems based on experimental data from NMR and small-angle neutron and X-ray scattering (SANS/SAXS) combined with in silico modeling. We have developed the Virtual NMR Spectrometer, a computer tool for learning/teaching NMR and for accurate prediction of the outcome of modern NMR experiments.


